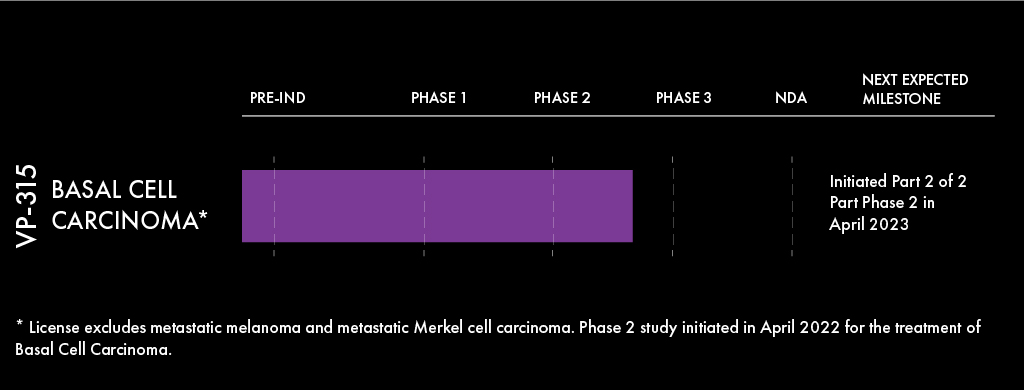
Pipeline VP-315
Skin cancer is the most common of all types of cancer. The American Cancer Society estimates that about 5.4 million basal and squamous cell skin cancers are diagnosed in the US each year. Over ¾ of these skin cancers are basal cell carcinoma (BCC), which is the most common malignancy in humans. Increasing age and sun exposure are associated with an increased risk for BCC and may contribute to the increasing incidence of this tumor type. Commonly used office procedures to treat BCC include surgical excision, Mohs micrographically controlled surgery, and electrodessication and curettage, although these are invasive procedures which can be associated with scarring.
Verrica has in-licensed LTX-315, an oncolytic peptide administered directly to the tumor to
induce immunogenic cell death, which may offer a non-surgical option for patients suffering from skin cancer. Verrica has an exclusive worldwide license to develop and commercialize LTX-315 for dermatologic oncology indications and intends to focus initially on basal cell and squamous cell carcinomas as the lead indications for development. LTX-315 has demonstrated positive tumor-specific immune cell responses in multi-indication Phase I/II oncology trials.
References: American Cancer Society. Facts & Figures 2020. American Cancer Society.
Atlanta, Ga. 2020.